Parasite Control
Acanthocephala or thorny-headed worms are endoparasites with a complex life cycle and a worldwide distribution. Acanthocephala also parasitize human livestock and have recently gained importance as a pest in fish farming. To provide the basis for effective and specific control of these parasites, we are investigating their evolution, phylogenetic relationships and development using genomic, transcriptomic and proteomic data. Our data reflect a complex energy strategy with simultaneous respiration and fermentation in acanthocephalans (see figure below after Mauer et al. 2020). Also, we found females to be more affected by living in an unsuitable host than than males (Schmidt et al. 2022a). Furthermore, we were able to identify acanthocephalan proteins as targets for their effective and specific control in aquaculture. This included the identification of compounds that can now be tested in the acanthocephalan model (Schmidt et al. 2022b). We are currently continuing to study the genomic architecture of Acanthocephala, the development of transcriptomes and the reproduction of Acanthocephala.

Phylogeny and Evolution of Thorny-headed Worms
We investigate the phylogenetic relationships and evolution of endoparasitic thorny-headed worms (Acanthocephala) and wheel animals based on molecular data. Juvenile acanthocephalans mature in the body cavities of insects, myriapods and crustaceans (intermediate hosts), while adult worms live and reproduce in the intestinal tracts of vertebrates including primates (definitive hosts). Our molecular analyses suggest that the acanthocephalan endoparasitism evolved from free-living ancestors via an epizoic stage living on a mandibulate host. According to this, expansion of the livecycle by a second host of higher trophic level took place subsequently. This scenario may serve as a model for the evolution of complex parasitic lifecycles (see Wey-Fabrizius et al. 2014).
Phenotype Evolution in Light of Mitochondrial Phylogeny
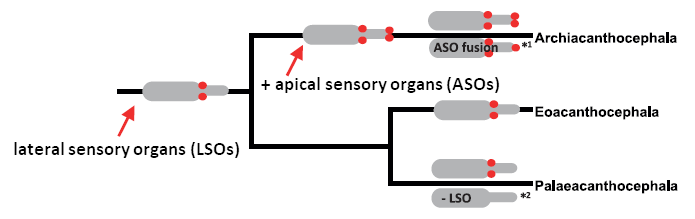
Our investigations additionally take into account morphological data. For example, we mapped the presence-absence of acanthocephalan sensory structures on a mitochondrial phylogeny. This revealed that paired lateral sensory organs (LSOs) most probably evolved in the acanthocephalan stem line while paired apical sensory organs (ASOs) should have emerged inside the acanthocephalan tree, followed by fusion of these paired organs to an unpaired structure in archiacanthocephalan evolution. A reduction of lateral sensory organs might have occured in acanthocephalan evolution too. Thus, acanthocephalan sensory organs illustrate the enormous phenotypic plasticity of endoparasitic acanthocephalans (see below Figure modified after Weber et al. 2013).
You may also visit: